Hindered Amine Light Stabilizers (HALS)
The "Radical Scavengers" of Polymer Science — Professional Formulation Solutions

In many plastic and polymer applications, UV-induced degradation is an inevitable challenge. Products such as automotive exterior parts, outdoor furniture, construction materials, and agricultural films often exhibit fading, chalking, embrittlement, or loss of mechanical strength after prolonged exposure to sunlight. These changes not only compromise aesthetics but also significantly shorten the service life of the material.
To enhance weatherability, formulation engineers typically incorporate light stabilizers. Among these, Hindered Amine Light Stabilizers (HALS) represent the most critical and widely utilized light stabilization technology in the polymer industry today.
What are Hindered Amines?
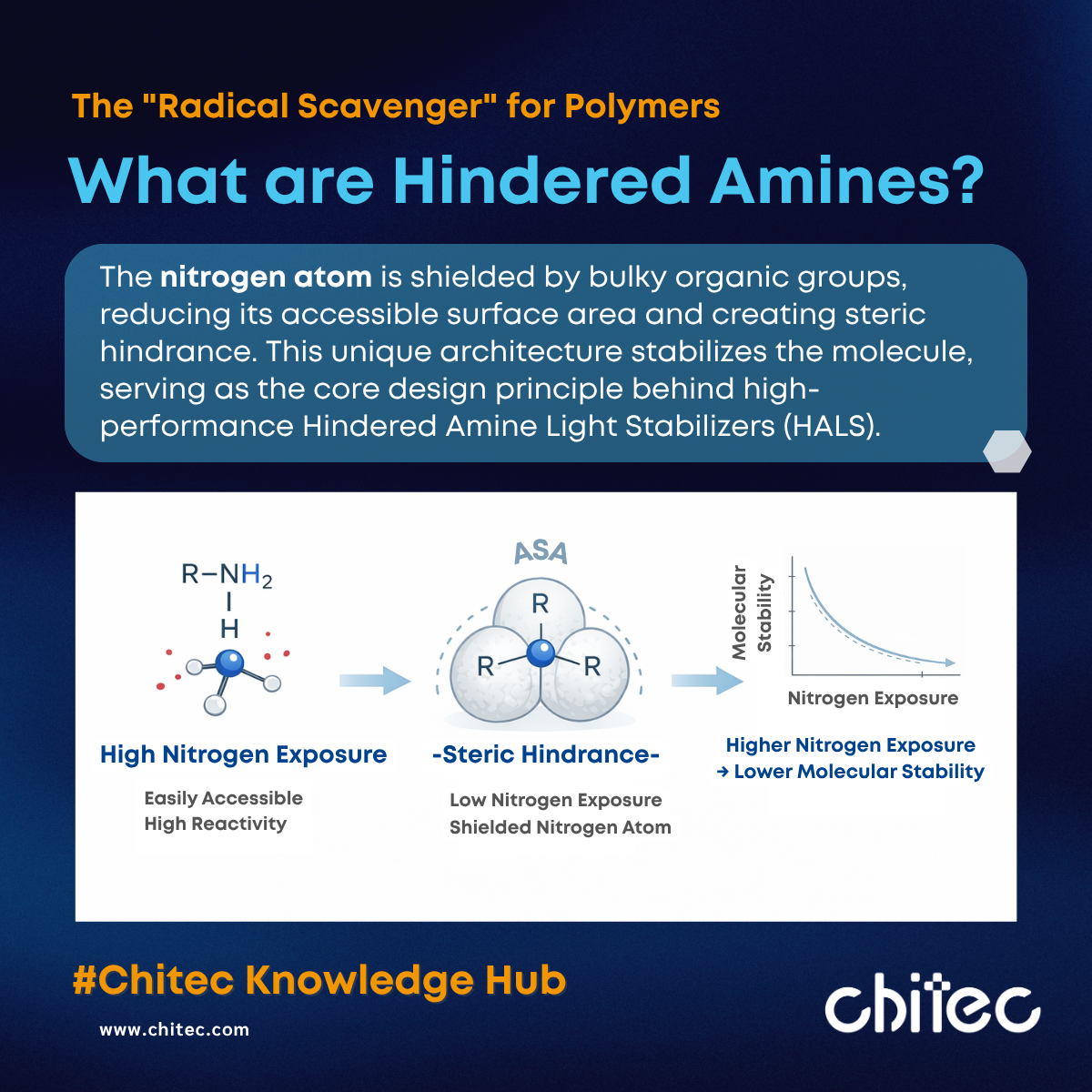
To understand HALS, one must first understand the concept of a "hindered amine." These are a class of amine compounds with a specific molecular architecture where the nitrogen atom is surrounded by bulky organic groups. This creates a crowded environment known as steric hindrance.
Due to these large substituents, undesirable chemical side reactions are blocked, while the specific reactive capacity required for stabilization is preserved. This unique structure grants hindered amines excellent chemical stability and allows them to function continuously during the photo-oxidation process.
How Does UV Radiation Cause Degradation?
When plastics or coatings absorb UV energy, chemical bonds within the material can break, generating highly reactive molecules called free radicals. These radicals react rapidly with oxygen to form new radicals, triggering a cascading chain reaction.
As this process continues, polymer chains are progressively severed, leading to a drop in molecular weight. This ultimately results in physical failure, such as brittleness, surface chalking, or discoloration. This entire process is known as photo-oxidation. Without an appropriate stabilization mechanism, the radical reaction spreads exponentially, drastically accelerating the aging process.
How Do HALS Capture Free Radicals?
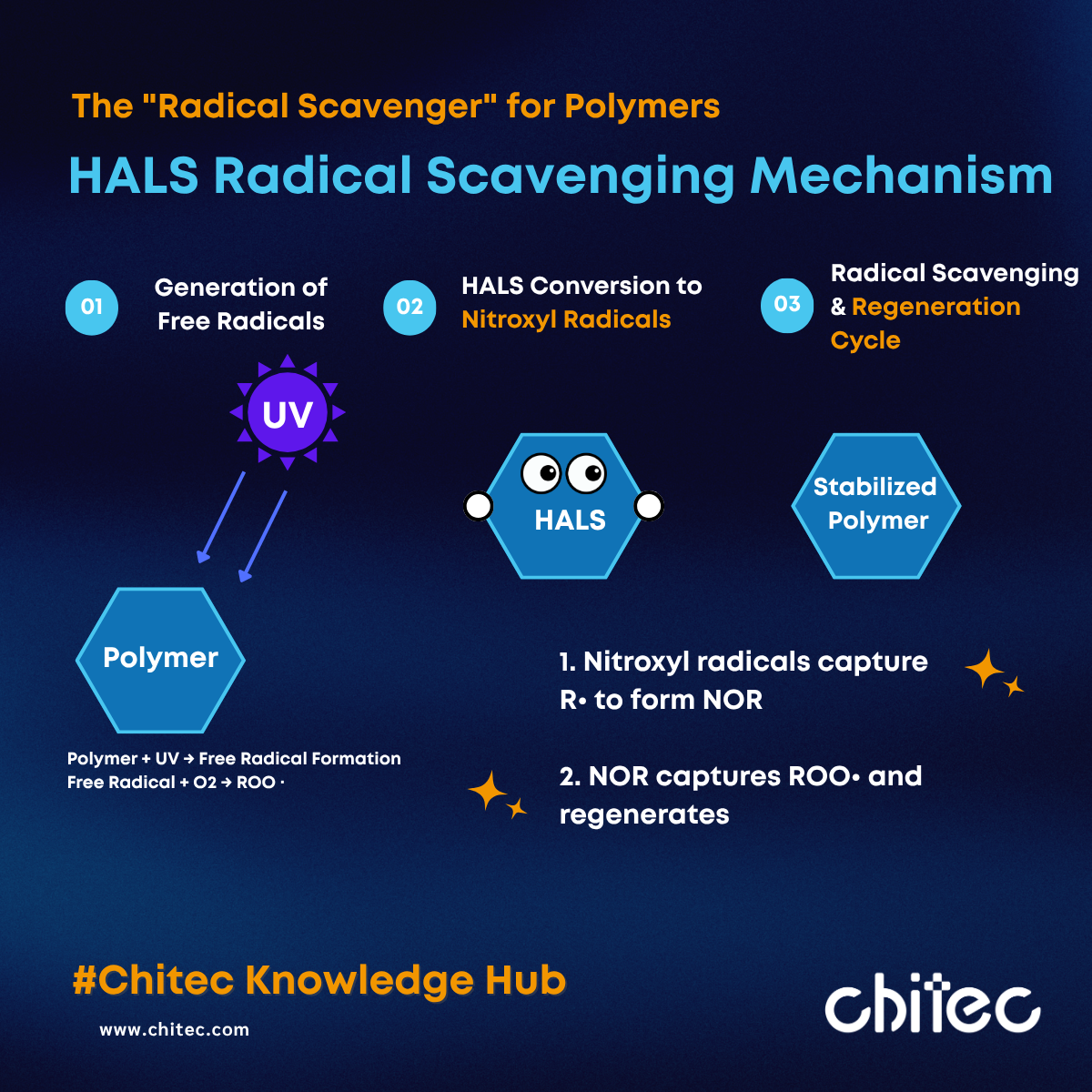
The mechanism of HALS differs fundamentally from that of UV Absorbers. HALS do not protect materials by absorbing UV radiation; instead, they intervene after radicals have formed by inhibiting the chain reaction through the well-known Denisov Cycle.
Phase 1: Formation of the Nitroxyl Radical
In the early stages of photo-oxidation, HALS is converted into its active form: the Nitroxyl Radical. This radical preferentially captures carbon-centered radicals (alkyl radicals) to form a stable alkoxyamine intermediate (NOR).
Phase 2: The Regenerative Cycle
The NOR structure then reacts with highly destructive peroxyl radicals. In this step, the peroxyl radicals are neutralized into stable products, while the NOR species is regenerated back into a nitroxyl radical. This cyclic mechanism allows a single HALS molecule to capture radicals repeatedly, providing long-term protection even at low loading levels.
Synergistic Effects: Why HALS is Rarely Used Alone
In commercial formulations, HALS is typically paired with other stabilizers to create a comprehensive protection system. The most common pairing is with UV Absorbers (UVAs). While UVAs absorb UV energy and dissipate it as harmless heat (preventing radical formation), HALS scavenge the radicals that have already been generated.
Furthermore, HALS is often used alongside Antioxidants. Antioxidants primarily protect the polymer from thermo-oxidation during high-temperature processing (such as extrusion or injection molding), while HALS provide long-term protection against photo-oxidation during the product's outdoor service life. This multi-layered approach ensures material stability from processing to end-use.
HALS Structures: N-H, N-R, and N-OR

HALS are categorized into three main types based on the substituent on the nitrogen atom. These structural differences dictate their reactivity and compatibility with various resin systems.
N-H Type HALS
The conventional structure where a hydrogen atom is attached to the nitrogen. These are highly efficient at radical scavenging and are widely used in polyolefins (PE, PP). However, they can be sensitive to acidic environments.
N-R Type HALS
The hydrogen is replaced by an organic substituent (R), such as a methyl group. This substitution increases molecular stability and reduces unwanted side reactions, making them ideal for high-temperature processing in engineering plastics.
N-OR Type HALS (Alkoxyamines)
The nitrogen is bonded to an alkoxy group (OR). Since they are non-basic, they do not react with acidic species. This makes them the preferred choice for coatings or formulations containing acidic flame retardants or agrochemicals.
Other High-Performance HALS:

Applications: From Plastics to Coatings
HALS is essential for any application requiring long-term outdoor durability, including automotive trim, roofing membranes, and greenhouse films. In the coatings industry, HALS helps maintain gloss and color stability, preventing chalking in harsh environments.


建議您使用以下瀏覽器觀看本網站,
要下載瀏覽器,請直接點擊以下:以獲得最佳瀏覽效果。